Workplace air -- determination of mercury and inorganic mercury compounds -- method by cold-vapour atomic absorption spectrometry or atomic fluorescence spectrometry.
ISO
Geneva: International Organization for Standardization, ISO 17733:2004, 2004 Nov; :1-51
20031310
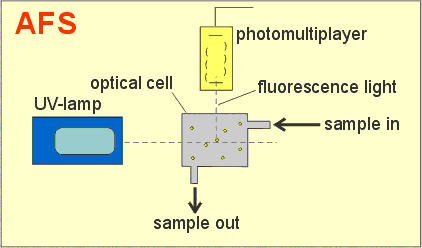
Cold Vapor Atomic Fluorescence (CVAF) is a powerful technique based on detecting fluorescence light emitted by the sample. The CVAF technique is an extre. The mercury analyzers as a detectors use a cold vapor atomic fluorescence spectrometry method (CVAFS). In the CVAFS, light from small mercury vapor lamp is.
ISO 17733:2004 specifies a procedure for determination of the time-weighted average mass concentration of mercury vapour and inorganic mercury compounds in workplace air. Mercury vapour is collected on a solid sorbent using either a diffusive badge or a pumped sorbent tube. Particulate inorganic mercury compounds, if present, are collected on a quartz fibre filter. Samples are analysed using either cold vapour atomic absorption spectrometry (CVAAS) or cold vapour atomic fluorescence spectrometry (CVAFS) after acid dissolution of the mercury collected. ISO 17733:2004 is applicable to the assessment of personal exposure to mercury vapour and/or particulate inorganic mercury compounds in air for comparison with long-term or short-term exposure limits for mercury and inorganic mercury compounds and for static (area) sampling. The lower limit of the working range of the procedure is the quantification limit. This is determined by the sampling and analysis methods selected by the user, but it is typically in the range 0,01 g to 0,04 g of mercury (see 13.1). The upper limit of the working range of the procedure is determined by the capacity of the diffusive badge, sorbent tube or filter used for sample collection, but it is at least 30 g of mercury (see 13.2). The concentration range of mercury in air for which ISO 17733:2004 is applicable is determined in part by the sampling method selected by the user, but it is also dependent on the air sample volume. The diffusive badge method is not applicable to measurements of mercury vapour when chlorine is present in the atmosphere, e.g. in chloralkali works, but chlorine does not interfere with the pumped sorbent tube method (see 13.11.1). Gaseous organo-mercury compounds could cause a positive interference in the measurement of mercury vapour (see 13.11.2). Similarly, particulate organo-mercury compounds and gaseous organo-mercury compounds adsorbed onto airborne particles could cause a positive interference in the measurement of particulate inorganic mercury compounds (see 13.11.3).
Analytical-methods; Analytical-processes; Workplace-monitoring; Air-sampling; Air-sampling-techniques; Mercury-compounds; Atomic-absorption-spectrometry
7439-97-6
20041118
Book or book chapter

2005
DART
Workplace air -- determination of mercury and inorganic mercury compounds -- method by cold-vapour atomic absorption spectrometry or atomic fluorescence spectrometry
OH
- Barghigiani C & Ristori T (1995) Preliminary study on mercury uptake by Rosmarinus officinalis L. (Rosemary) in a mining area (Mt. Amiata, Italy). Bull. Environ. Contam. Toxicol. 54: 519–525Google Scholar
- Barghigiani C & Bauleo R (1992) Mining area environmental mercury assessment using Abies alba. Bull. Environ. Contam. Toxicol. 49: 31–36Google Scholar
- Barghigiani C, Bargagli R, Siegel BZ & Siegel SM (1990) A comparative study of mercury distribution on the Aeolian volcanoes, Vulcan and Stromboli. Water Air Soil Poll. 53: 179–188Google Scholar
- Bargagli R, Iosco FP & Barghigiani C (1991) Assessment of mercury dispersal in an abandoned mining are by soil and lichen analysis. Water Air Soil Poll. 56: 219–233Google Scholar
- Carpi A, Weinstein LH & Ditz DH (1994) Bioaccumulation of mercury by Sphagnum moss near a municipal solid waste incinerator. Air & Waste 44: 669–672Google Scholar
- CEM (1991) Microwave Sample Preparation: Applications Manual. CEM Corporation, Matthews, NCGoogle Scholar
- Fischer RG, Rapsomanikis S & Andreae MO (1995) Bioaccumulation of methylmercury and transformation of inorganic mercury by macrofungi. Environ. Sci. Technol. 29: 993–999Google Scholar
- Fitzgerald WF & Gill GA (1979) Subnanogram determination of mercury by two-stage gold amalgamation and gas phase detection applied to atmospheric analysis. Anal Chem. 51: 1714–1720Google Scholar
- Iverfeldt A (1991) Mercury in forest canopy throughfall water and its relation to atmospheric deposition. Water Air Soil Poll. 56: 553–564Google Scholar
- Keeler G, Glinsorn G & Pirrone N (1995) Particulate mercury in the atmosphere: its significance, transport, transformation and sources. Water Air Soil Poll. 80: 159–168Google Scholar
- Kojo M-R & Lodenius M (1989) Cadmium and mercury in macrofungi – Mechanisms of transport and accumulation. Angew. Botan. 63: 279–292Google Scholar
- Kovalevskii AL (1986) Mercury-biogeochemical exploration for mineral deposits. Biogeo. 2: 211–220.Google Scholar
- Lindberg SE (1997) Mercury Air/Surface Exchange (MASE) Project – Annual Summary Report. Electric Power Research InstituteGoogle Scholar
- Lindberg SE (1996) Forests and the global biogeochemical cycle of mercury: The importance of understanding air/vegetation exchange processes. In: Baeyens W, Ebinghaus R & Vasiliev O. (Eds) Global and Regional Mercury Cycles: Sources, Fluxes, and Mass Balances (pp 359–380). Kluwer Academic Publishers, Dordrecht, the NetherlandsGoogle Scholar
- Lindberg SE, Jackson DR, Huckabee JW, Janzen SA, Levin MJ & Lund JR (1979) Atmospheric emission and plant uptake of mercury from agricultural soils near the Almaden Mercury Mine. J. Environ. Qual. 8: 572–578Google Scholar
- Moore TR, Bubier JL, Heyes A & Flett RJ (1995) Methyl and total mercury in boreal wetland plants, experimental lakes area, northwestern Ontario. J. Environ. Qual. 24: 845–850Google Scholar
- Munthe J, Hultberg H & Iverfeldt A (1995) Mechanisms of deposition of methylmercury and mercury to coniferous forests. Water Air Soil Poll. 80: 363–371Google Scholar
- Olmez, I (1995) personal communicationGoogle Scholar
- Rasmussen PE (1994) Mercury in vegetation of the Precambrian shield. In: Watras CJ & Huckabee JW (Eds) Mercury as a Global Pollutant (pp 417–425). Lewis Publishers, Palo AltoGoogle Scholar
- Rasmussen PE, Mierle G & Nriagu JO (1991) The analysis of vegetation for total mercury. Water Air Soil Poll. 56: 379–390Google Scholar
- Rea AW, Keeler GJ & Scherbatskoy T (1996) The deposition of mercury in throughfall and litterfall in the Lake Champlain Watershed: A short-term study. Atmos. Environ. 30: 3257–3263Google Scholar
- Shaw BP & Panigrahi AK (1986) Uptake and tissue distribution of mercury in some plant species collected from a contaminated area in India: Its ecological implications. Arch. Environ. Contam. Toxicol. 15: 439–446Google Scholar
- Siegel BZ, Siegel SM & Horsky SJ (1984) Equisetum plants and the cycling of mercury at Mount St. Helens. Eviron. Sci. Technol. 18: 179–181Google Scholar
- Stokes PM, Dreier SI, Farkas MO & McLean RAN (1983) Mercury accumulation by filamentous algae: A promising biological monitoring system for methyl mercury in acid-stressed lakes. Environ. Poll. 5:255–271Google Scholar
